
New York City Health Department Annual Tuberculosis Summary, 2019

>> MISSION: The New York City (NYC) Health Department aims to
prevent the spread of tuberculosis (TB) and eliminate it as a public
health problem in NYC.
GOALS
Identify all individuals with suspected
or conrmed TB disease and ensure
their appropriate treatment, ideally
on directly observed therapy (DOT).
1
Ensure that individuals at high risk
for progression from latent TB
infection to TB disease complete
treatment and do not develop disease.
2
CORE ACTIVITIES
To fulll its mission and core goals related to TB care, prevention and elimination, the Health Department
performs a number of integrated activities in collaboration with local health care providers, laboratories,
community partners and others. With a focus on public health and the clinical care needs of patients and
their families, these activities support effective TB care and control in NYC.
CLINICAL CARE
LABORATORY
TESTING
CONTACT
INVESTIGATION
STRATEGIC
DATA USE
TREATMENT
CASE
MANAGEMENT
OUTBREAK DETECTION
AND RESPONSE
REPORTING AND
SURVEILLANCE
MEDICAL
CONSULTATION
OUTREACH AND
EDUCATION
TABLE OF CONTENTS
INTRODUCTION
Tuberculosis in New York City, 2019 ................................................................................................................. 4
Letter from the Assistant Commissioner ..........................................................................................................5
CORE ACTIVITIES
Surveillance.......................................................................................................................................................7
Clinical Care and Treatment .............................................................................................................................8
Medical Consultation ........................................................................................................................................9
Case Management ............................................................................................................................................9
Drug Susceptibility Testing, Genotyping and Whole Genome Sequencing ....................................................11
Outreach and Education ................................................................................................................................ 12
Program Evaluation ........................................................................................................................................ 12
Funding and Administration ........................................................................................................................... 13
Research ........................................................................................................................................................ 13
Spotlight on Coalition-Building, Equity and Internal Reform ..........................................................................15
PROFILE OF TUBERCULOSIS CASES, 2019
Tuberculosis in New York City ........................................................................................................................ 18
Age and Sex .....................................................................................................................................................19
Birth in the United States .............................................................................................................................. 20
Country of Birth ...............................................................................................................................................21
Tuberculosis in New York City Neighborhoods ............................................................................................... 22
Drug Resistance ..............................................................................................................................................24
Disease Site, Culture Status and Co-morbidities .......................................................................................... 26
Mortality, Treatment Completion and Indicators ............................................................................................27
Contact Investigation in Non-Household Settings ......................................................................................... 28
Whole Genome Sequencing and New York City Clusters .............................................................................. 29
Select Demographic, Social and Clinical Charateristics by Birth in the United States ................................. 30
APPENDICES
Historical Tuberculosis Data, New York City .................................................................................................. 30
Tuberculosis Reporting Requirements ........................................................................................................... 33
Tuberculosis Reporting Data .......................................................................................................................... 35
Historical Tuberculosis Data .......................................................................................................................... 36
Resources for Providers and the Public ..........................................................................................................37
Technical Notes .............................................................................................................................................. 38
New York City Health Department Chest Clinics ........................................................................................... 39
ABOUT THIS REPORT: This report provides surveillance data and summaries of core program activities for calendar year 2019.
The data reect the most complete information available as of January 15, 2020. For additional details on the use of denominators
and denitions in this report, please see Technical Notes (page 36).
PREPARED BY: Shama Ahuja, PhD, MPH; Jillian Knorr, MPH; Jyotsna Ramachandran, MPH; Jeanne Sullivan Meissner, MPH;
Lisa Trieu, MPH
PROGRAM CONTENT PROVIDED BY: Joseph Burzynski, MD, MPH; Magali Calderon, MS; Christine Chuck, MPA; Felicia Dworkin, MD;
Hannah Jordan, MD, MPH; Michelle Macaraig, DrPH; Herns Modestil, BS; Diana Nilsen, RN, MD; Farah Parvez, MD, MPH;
Shaila Rao, EdD, MPH; Errol Robinson, MPA
SUGGESTED CITATION: New York City Department of Health and Mental Hygiene. Bureau of Tuberculosis Control Annual Summary,
2019. Queens, NY. 2020.
COVER ART: "Breathe" by Dr. Sneha

100%
Proportion of
neighborhoods
1
with at least
one TB case
84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 11 12 13 14 15 16 17 18 19
71%
of TB cases among
the U.S.-born were
among Hispanic or
non-Hispanic Black
patients
62%
of TB cases occurred
among people who
were 45 years of
age or older
IN NEW YORK CITY, 2019
566
Number of TB
cases veried
in NYC in 2019
60%
of TB cases occurred
among males
PATIENT CHARACTERISTICS
6.9
NYC citywide
TB rate per
100,000 people
Number of cases
Rate per 100,000
Year
45 to 64
31%
65 and older
31%
0 to 17
3%
18 to 44
35%
Asian
3%
1
Hispanic
26%
1
NYC HEALTH DEPARTMENT TB SERVICES AND KEY ACTIVITIES
The NYC Health Department performs a range of TB services and activities in collaboration with local health care providers, laboratories,
community partners and others. Health Department services are provided free of charge to ALL PATIENTS, regardless of the patient's
immigration status, insurance status or ability to pay. For a list of Health Department chest clinics, see page 37.
2% INCREASE
in the number of TB
cases from 2018-2019
1. Among U.S.-born patients (n=91). Two patients
had unknown race/ethnicity.
Multiple/Other
7%
1
CLINICAL CARE LABORATORY
TESTING
CONTACT
INVESTIGATION
STRATEGIC
DATA USE
TREATMENT CASE
MANAGEMENT
OUTBREAK
DETECTION AND
RESPONSE
REPORTING AND
SURVEILLANCE
MEDICAL
CONSULTATION
OUTREACH AND
EDUCATION
COUNTRY OF BIRTH
70
Number of countries of
birth represented among
patients with TB disease
Born in the United States (U.S.) 16%
Not U.S.-born 84%
MULTIDRUG RESISTANCE
Median number of drugs
to which there was known
drug resistance among
MDR-TB cases
(range 3-10)
7
11
Number of patients
diagnosed in 2019 who
had multidrug-resistant
TB (MDR-TB)
1
1. Dened as resistance to at
least isoniazid and rifampin.
Female Male
Non-Hispanic
White
16%
1
Non-Hispanic Black
45%
1
TB IN NYC NEIGHBORHOODS
1
15
Number of neighborhoods
1
with a TB rate higher than
the 2019 NYC citywide rate
1. Dened by United Hospital Fund neighborhood
designation (n=42). 2. Rates are per 100,000.
At or below
provisional
national rate
2
(0.7 to 2.7)
Above citywide rate
2
(7.0 to 19.9)
At or below citywide
rate
2
(2.8 to 6.9)
40% 60%

March 2020
DEAR COLLEAGUES,
Last year was full of promising steps in the ght against TB, including advances in the development and
implementation of new diagnostic, treatment and prevention tools; continued coalition-building; renewed
energy for advocacy; and collective efforts toward TB elimination.
The incidence of TB has remained steady in NYC, with 566 cases of active TB disease counted in 2019.
Among those, 11 cases involved a multidrug-resistant TB (MDR-TB) strain. At a disease rate of 6.9 per
100,000, more than twice the national rate, we must approach TB treatment and prevention from new
angles and with improved focus.
In 2019, the NYC Health Department continued to move toward a more patient-centered approach to TB
care. We expanded social support services for patients and their families. We continued to make directly
observed therapy (DOT) easier and more accessible for patients by expanding the use of video-based DOT.
We developed a contact case management unit, which enhanced our ability to provide blood-based TB
testing and HIV testing to contacts in household settings. In our clinics, we maintained extended hours and
weekend clinical services. We also continued to use whole genome sequencing and other molecular tests to
more rapidly identify drug resistance and ensure that patients are placed on appropriate treatment sooner.
In order to be successful in our ght against TB, we need to maintain strong ties with external partners. To
that end, we strengthened our collaboration with interdisciplinary stakeholders through the Coalition for
a TB-Free NYC. In order to think critically about our treatment of drug-resistant TB, we met with local TB
experts, advocates and patient representatives to discuss how new MDR-TB treatment guidelines released
by the World Health Organization (WHO) might inuence treatment. The outcome of this meeting was to
preferentially adapt MDR-TB regimens to avoid medications administered by injection. These regimens
include new drugs that are effective, well-tolerated and avoid the discomfort and potential for serious side
effects associated with injectable TB medications.
TB remains a formidable challenge in NYC. Yet the Health Department remains undaunted and committed
to our mission. As always, we feel fortunate to be able to work closely with our many partners to ensure that,
collectively, we can continue to provide the best possible care to our patients and communities as we work
toward a TB-free NYC.
Sincerely,
Joseph N. Burzynski, MD, MPH
Assistant Commissioner, Bureau of Tuberculosis Control
New York City Department of Health and Mental Hygiene
>> LETTER FROM THE ASSISTANT COMMISSIONER
5 New York City Health Department Annual Tuberculosis Summary, 2019

CORE ACTIVITIES

Conrmed TB cases
were veried by the
Health Department.
People with suspected
TB disease were
reported to the Health
Department.
Children younger than
5 years of age with
TB infection were
reported to the
Health Department.
Facilities reported at
least one TB case;
nearly half of all cases
were reported by one
of 14 facilities.
CORE ACTIVITIES
TB is an airborne, infectious disease caused by a bacteria. TB has
two stages: active TB disease and latent TB infection (LTBI). TB is
treatable and preventable, but without effective treatment, TB may
lead to serious illness and death.
The NYC Health Department performs a variety of integrated activities
to address TB disease and LTBI. These include surveillance, clinical
care and treatment, medical consultation, case management, contact
investigation, coordination of laboratory tests (e.g., drug susceptibility
testing and genotyping), outbreak detection and response, outreach,
program evaluation and research. These activities support effective,
patient-centered TB care, control and prevention in NYC.
SURVEILLANCE
Health care providers and laboratories are required to report to the
Health Department:
1. All patients with conrmed TB disease
2. Anyone suspected of having TB disease
3. Children younger than 5 years of age with a positive test for TB
infection and related chest radiograph ndings and treatment
information
4. Results of any blood-based test for TB infection, regardless of
patient age (laboratories only)
The Health Department reviews submitted reports for completeness
and timeliness and determines whether patients are eligible for case
management. The Health Department maintains an electronic TB
surveillance registry and case management system (Maven version
5.4.3.1, Conduent Public Health Solutions, Florham Park, NJ) that
includes information for all reported patients and individuals exposed
to TB. These data are used to conduct case management; ensure
treatment completion; monitor trends; detect, investigate and respond
to outbreaks; prepare surveillance reports; report aggregated data to
the New York State Department of Health (NYSDOH) and the Centers
for Disease Control and Prevention (CDC); identify reporting and data
quality issues; and inform programmatic decision-making.
7 New York City Health Department Annual Tuberculosis Summary, 2019
566
3,191
109
93
>> 2019 BY THE NUMBERS
SURVEILLANCE:
>> HEALTH CODE CHANGE
FOR LTBI REPORTING:
In 2019, the NYC Health
Code was amended to
require laboratories to
report results of all
blood-based tests for
TB infection to the Health
Department, regardless of
patient age or test result.
This change became
effective Nov. 15, 2019.
>> For more information
about NYC TB reporting
requirements and related
resources, see pages 32-33.
>> CORE ACTIVITIES

Patients conrmed
with TB disease in
2019 received care at
a Health Department
chest clinic.
Patients with an
MDR-TB strain
received treatment,
care and case
management
through the Health
Department, including
11 patients newly
diagnosed with an
MDR-TB strain in
NYC and 10 patients
initially veried as TB
cases outside of NYC.
Patients received
bedaquiline; 15
patients received
clofazimine.
Immigrants and
refugees arriving in
NYC with a Class B
designation were
notied to the Health
Department; 1,154
(91%) were eligible
for evaluation; 703
(61%) were evaluated
as of Jan. 15, 2020.
284
46
18
1,266
>> 2019 BY THE NUMBERS
CLINICAL CARE & TREATMENT:
CLINICAL CARE AND TREATMENT
The Health Department is a leading provider of TB care in NYC. TB
services are available at four Health Department chest clinics located
in The Bronx, Brooklyn, Manhattan and Queens. Physicians working at
the chest clinics are specialists in internal, preventive, and pulmonary
medicine, pediatrics and infectious disease.
The Health Department provides TB diagnostic services, including
testing for TB infection (using blood-based QuantiFERON-TB Gold Plus
[QFT] test and tuberculin skin test [TST]), sputum induction, laboratory
tests, medical evaluation, chest radiographs, treatment for TB disease
and LTBI and directly observed therapy (DOT) services. The majority
of patients evaluated and treated at Health Department chest clinics
are referred by NYC health care providers, other health departments
or social service providers. Health Department staff refer patients to
other medical professionals for further evaluation and treatment of
non-TB related conditions.
OBTAINING MEDICATIONS WITH LIMITED AVAILABILITY FOR THE TREATMENT
OF MDR-TB: Health Department staff can assist with obtaining certain
medications that are available under limited circumstances, including
delamanid, bedaquiline and clofazimine. Clofazimine and delamanid
require the submission of a Single Patient Investigational New Drug
application to the Food and Drug Administration and to the Health
Department's Institutional Review Board for approval.
EVALUATION OF NEWLY ARRIVED IMMIGRANTS AND REFUGEES: People who
are applying for permanent U.S. immigration status and refugee status
are screened for TB as part of their overseas medical examination.
If the pre-immigration examination nds clinical diagnosis of TB, a
Class A designation is given and the applicant is not allowed to travel
until treatment is completed or the patient is no longer infectious.
If ndings suggest non-infectious TB, the applicant is given a Class
B designation and travel clearance and the applicant's destination
city is notied by CDC. The destination city then noties that individual
of the need for TB re-evaluation. The Health Department follows up
with all immigrants and refugees who arrive in NYC with Class B status.
The majority come to a Health Department chest clinic for evaluation.
For more information about these drugs or for help obtaining them,
please call the TB HOTLINE at 844-713-0559.
8 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

Patients received
case management
services, including 566
patients with newly
conrmed TB disease,
1,375 patients with
suspected TB disease,
412 patients with TB
diagnosed before
2019 and 99 patients
with TB initially veried
outside of NYC.
Eligible patients with
conrmed TB disease
were enrolled in DOT
through the Health
Department or another
health care provider.
127 patients were
enrolled exclusively
in face-to-face DOT;
266 received some
or all observations
through vDOT.
Contacts were
identied for 463
potentially infectious
patients; 2,365 (70%)
were evaluated as of
Jan. 15, 2020; 372
(16%) had a new
positive TB test result.
2,452
393
3,376
>> 2019 BY THE NUMBERS
CASE MANAGEMENT:
HIV TESTING SERVICES: Health Department staff provide opt-out rapid
HIV testing services to every eligible patient at Health Department
chest clinics and refer patients who have HIV infection to health care
providers who specialize in HIV care. In 2019, the Health Department
began offering rapid HIV testing to contacts in household settings.
Anonymous HIV testing services are available at all chest clinics
independent of need for TB services.
MEDICAL CONSULTATION
Health Department TB medical consultants are physicians with years
of experience treating TB disease and LTBI. They provide expert
consultation to community-based care providers and others regarding
TB diagnosis; hospital discharge planning; TB treatment (including
treatment of MDR-TB, adverse reactions to TB drugs and treatment
completion); contact investigation; infection control; and other TB-
related public health concerns. Recommendations are based on these
physicians’ professional experience and Health Department policies,
which are informed by guidelines from the CDC, the American Thoracic
Society, the Infectious Diseases Society of America, the National TB
Controllers Association and WHO. Medical consultants also conduct
TB rounds and give medical talks throughout NYC.
CASE MANAGEMENT
The Health Department provides case management activities for NYC
residents diagnosed with or suspected of having TB disease and their
contacts, regardless of where the patient receives their TB care. Case
management includes patient interviews, TB education, chart reviews,
contact investigation, DOT and coordination with community providers
to ensure optimal TB treatment and care. Health Department staff
conduct home assessments to identify contacts and to determine
whether infectious TB patients can be discharged from the hospital.
The Health Department also coordinates with colleagues in other
jurisdictions to ensure continuity of care for patients with conrmed TB
disease and contacts who work or live outside of NYC. Case managers
perform monthly monitoring for treatment adherence and locate
patients who have signicant lapses in medical appointments or
medication and help them return to medical supervision.
For consultation related to the management and treatment of TB,
including MDR-TB, please call the TB HOTLINE at 844-713-0559.
9 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

>> 2019 SPOTLIGHT ON...
DIRECTLY OBSERVED THERAPY: DOT is the standard of care for patients
with suspected or conrmed TB disease in NYC, regardless of where
they receive TB care. During DOT, a health care worker observes a
patient ingesting their anti-TB medications. The Health Department
provides face-to-face DOT services at all chest clinics and at homes,
worksites and other locations as requested by the patient. The Health
Department also provides video DOT (vDOT), which facilitates continuity
of DOT outside traditional business hours and when patients travel.
Face-to-face DOT is also available through three NYC Health+Hospitals
facilities: NYC Health+Hospitals/Elmhurst, NYC Health+Hospitals/
Kings County and NYC Health+Hospitals/Bellevue.
CONTACT INVESTIGATION: The Health Department conducts TB contact
investigations in household, social and other congregate settings
(e.g., worksites and schools). During contact investigation, Health
Department staff identify and evaluate individuals who were exposed
to infectious TB patients, ensure appropriate treatment among
contacts who are diagnosed with TB disease or LTBI, determine whether
transmission occurred and assess whether further testing or other
intervention is needed. When TB exposures occur in health care
facilities, epidemiologists at the Health Department provide technical
guidance and assist with contact investigation at the site as needed.
SOCIAL SERVICE REFERRALS: Health Department staff identify and
address obstacles to care and unmet social service needs among
patients and their families whenever possible. These include concerns
about nances, housing, food security, unstable employment, school,
health insurance eligibility, access to health care services, immigration
status, language barriers, drug and alcohol use and mental health
issues. When these situations are identied, patients are referred to
a social worker, who facilitates referrals to social service resources.
REGULATORY ACTION: The Health Department has authority under the
New York City Health Code to mandate compulsory evaluation, DOT or
involuntary hospitalization for patients with infectious TB who are not
adherent to evaluation, isolation or treatment recommendations and
pose a public health risk.
To learn more about the DOT program at the Health Department or to
enroll a patient, please call 311.
TELEMEDICINE:
Starting in 2019, the Health
Department now offers
telemedicine services for
patients who initiate
treatment for LTBI at its
Corona Chest Clinic.
Eligible patients now have
the option to have follow-up
medical visits with a nurse
through video conferencing
instead of a traditional
in-person visit. The pilot
program will expand from
Corona to other sites in 2020
and telemedicine services
will be available to all eligible
patients being treated for
LTBI at a Health Department
chest clinic.
CONTACT CASE
MANAGEMENT UNIT:
In 2019, the Health
Department developed
a dedicated team to
coordinate evaluation of
household contacts. Health
Department staff in the
TB Contact Case Management
Unit conduct all home
assessments and provides
blood-based TB testing and
HIV testing to contacts in
household settings.
10 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

Culture-conrmed TB
cases had phenotypic
DST results available
(95%); among all cases,
molecular DST results
were available for
471 (83%) cases.
Culture-conrmed TB
cases had genotype
results available (83%);
438 (95%) cases had
WGS results available.
Instances of potential
false positive
laboratory results
were investigated;
9 investigations
conrmed a false
positive result.
436
384
37
>> 2019 BY THE NUMBERS
DST, GENOTYPING AND WGS:
DRUG SUSCEPTIBILITY TESTING, GENOTYPING
AND WHOLE GENOME SEQUENCING
The NYC Health Code mandates that a portion of the initial isolate from
all culture-positive TB patients be sent for drug susceptibility testing
(DST) and genotyping, which are performed at local, state and national
public health laboratories.
DRUG SUSCEPTIBILITY TESTING: DST identies drug resistance in TB
strains and informs clinical management and treatment for patients
with TB disease and their contacts. The NYC Public Health Laboratory
(PHL) performs phenotypic DST testing for rst-line and select second-
line TB drugs. Molecular-based laboratory tests are also being used
routinely at PHL, commercial laboratories, hospitals and other public
health reference laboratories. These tests rapidly conrm the presence
of Mycobacterium tuberculosis (M. tuberculosis) complex and can
provide information about the presence of mutations in specic genes
that are known to predict drug resistance.
GENOTYPING: Genotyping is a laboratory technique used to determine
the genetic pattern of M. tuberculosis strains. Genotyping results can
help identify whether TB strains are genetically related, which helps
the Health Department identify false positive laboratory results,
detect outbreaks and assess TB transmission. Possible instances of
contamination and potential false positive results are promptly and
systematically investigated to ensure patients are not placed on anti-TB
medications unnecessarily. Cases among patients with similar TB
strains are investigated to identify and interrupt TB transmission.
UNIVERSAL WHOLE GENOME SEQUENCING (WGS): In NYC, WGS enables
identication of the M. tuberculosis complex and species within it;
detection of genetic mutations associated with drug resistance; and
analysis of single nucleotide polymorphisms to characterize and
compare TB strains. The Health Department collaborates with the
NYSDOH Wadsworth Center and PHL to conduct WGS for all patients
with a positive culture for M. tuberculosis. NYS currently reports 17
mutations associated with resistance to 9 TB drugs. The CDC began
universal WGS for TB in May 2019 and reports results in conjunction
with outbreak investigation.
>> For more information about drug resistance among NYC cases in 2019, see pages
22-23. For more information about WGS and NYC clusters, see page 27.
Individuals were
tested for LTBI
during 5 community
health events
conducted in
collaboration with
community partners*
and made possible
with a grant from Stony
Wold-Herbert Fund.
106
OUTREACH:
* Including the Academy of Medical and Public
Health Services, the Ofce of Assembly Mem-
ber Felix Ortiz, the Charles B. Wang Community
Health Center, the Chinese Planning Council,
Mixteca, the Philippine Nurses Association,
Sapna NYC, Flushing YWCA and Queensboro
Community College
11 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

>> 2019 SPOTLIGHT ON...
OUTREACH AND EDUCATION
The Health Department engages various stakeholders to advance
efforts to detect, treat and prevent TB throughout NYC.
HEALTH DEPARTMENT STAFF: Educational materials developed by the
Health Department, the CDC and others are used to supplement staff
training, which is delivered by experts from the Health Department
and the Rutgers Global TB Institute.
HEALTH CARE PROVIDERS: Health care providers collaborate with the
Health Department in many capacities and are invited to discuss
TB testing, diagnosis and treatment through numerous channels.
Health Department experts participate in TB presentations and case
management conferences and provide clinical consultation at health
care facilities across NYC. The Health Department co-sponsors two
annual day-long educational events for health care providers and other
colleagues: a medical conference in honor of World TB Day; and a
conference to discuss advances in and applications of TB genotyping.
Bi-monthly trainings on TST are offered to physicians and nurses
from various agencies. In addition, Health Department staff regularly
consult with colleagues in local, national and international settings on
matters related to TB policy and practice.
COMMUNITIES: Communities with a high burden of TB are engaged as
partners in efforts to increase TB knowledge and inform care-seeking
behavior. In partnership with elected ofcials, community-based
groups and others, the Health Department delivers culturally and
linguistically appropriate educational messaging though community
events and print and electronic media. Community partners also
support TB screening efforts at health fairs and other testing events.
>> For more information about the Health Department's TB community events
or conferences, email [email protected]yc.gov.
PROGRAM EVALUATION
The Health Department uses local, state and national performance
indicators to assess program impact and success. These indicators
inform planning and policy decisions and help identify programmatic
issues and areas for improvement. Performance indicators and targets
are developed in coordination with Health Department partners and
funders, including CDC, NYSDOH and the NYC Mayor’s Ofce. Reports
In September 2019,
subject matter experts from
the Health Department, the
Mayor’s Ofce of Immigrant
Affairs, Safe Horizon, the New
York Immigration Coalition
and the United States
Citizenship and Immigration
Services came together to
provide essential updates on
the health of immigrants and
changes to the federal civil
surgeon technical instructions
for TB testing and reporting.
This event was co-sponsored
by the Rutgers Global TB
Institute, the Northeast
Caribbean AIDS Education
and Training Center, the U.S.
Citizenship and Immigration
Services and the Health
Department.
More than 100 federal
civil surgeons and other
providers from New York,
New Jersey, Pennsylvania
and Connecticut attended.
IMMIGRANT HEALTH CIVIL
SURGEON SYMPOSIUM:
For more information on
the updated technical
instructions for Civil
Surgeons, please visit
www.cdc.gov/
immigrantrefugeehealth/
12 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

include the Health Department’s Quality and Performance Review, a
quarterly report to NYSDOH, the CDC’s Annual Performance Report,
the CDC’s Aggregate Reports for TB Program Evaluation and the CDC's
National Tuberculosis Indicators Project.
>> For the most recent performance indicators, see page 25.
COHORT REVIEW: One of the Health Department’s primary tools for
evaluating its TB control program is the quarterly cohort review
process. Health Department staff review case management activities,
treatment status and data quality for all patients with conrmed TB
disease and their contacts four to six months after a patient's TB
diagnosis. Successes and challenges related to patient care and case
management are used to inform programmatic changes and identify
training needs.
FUNDING AND ADMINISTRATION
The Health Department receives city, state and federal funding for TB
care and control. Eighty-seven percent of the operating budget for the
Health Department's Bureau of TB Control supports personnel services
and 13% supports other-than-personnel services. These funds support
all TB prevention and control activities, from hiring staff to operating
Health Department chest clinics. Health Department staff ensure that
funds are allocated, monitored and utilized efciently.
RESEARCH
Health Department staff actively participate in research, including
epidemiologic studies, implementation science and clinical research.
This includes collaboration with the CDC TB Trials Consortium, which
conducts national and international studies to develop TB treatment
regimens. Health Department staff also participate in professional
organizations and TB advisory groups and coordinate the NYC TB
RESEARCH CONSORTIUM, which brings together Health Department,
academic and laboratory-based colleagues to collaborate on projects
focusing on TB in NYC.
CONFERENCES: NYC's TB data and expertise are shared at meetings
locally, nationally and internationally. In 2019, these included the 13th
Annual TB Medical Consultant’s Meeting, the 23rd Annual Conference
of The Union North America Region, the Advanced Molecular Detection
FIGURE 2: Bureau of Tuberculosis
Control funding distribution for
other-than-personnel services
by type, 2019
Laboratory testing services ..... 19%
Medications .............................. 18%
Patient support services .......... 15%
Electronic medical records ...... 15%
Other ........................................ 12%
Supplies/equipment ................ 10%
Chest radiograph services ....... 10%
FIGURE 1: Bureau of Tuberculosis
Control staff by job function,
2019 (n=222)
1,2
1. As of Jan. 15, 2020. 2. Excludes
13 non-Health Department personnel
Public health advisor ............... 41%
Data analyst/researcher ......... 14%
Other staff................................. 10%
Administrative professional ....... 9%
Nurse........................................... 8%
Physician ..................................... 7%
Administrator .............................. 6%
Other licensed professional ....... 4%
41%
14%
10%
9%
8%
7%
6%
4%
18%
15%
15%
12%
10%
10%
19%
13 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

>> STAFF PARTICIPATION IN
ADVISORY GROUPS AND
CONSORTIA, 2019
• Advisory Council for
the Elimination of TB
• CDC/Infectious
Disease Society of
America/American
Thoracic Society National
MDR-TB Guidelines
Writing Committee
• CDC TB Education and
Training Network
• CDC TB Program
Evaluation Network
• CDC TB Outbreak
Detection Workgroup
• CDC TB Trials Consortium
• Council of State and
Territorial Epidemiologists
• Maven Users Group
• National TB Controllers
Association: Board of
Directors; LTBI Reporting
Workgroup; National
Society of TB Clinicians;
Society for Epidemiology
in TB Control and Survey
Committee
• Northeastern TB Center
of Excellence for Training,
Education and Medical
Consultation Medical
Advisory Board
• International Union Against
TB and Lung Disease
conference, the Rutgers Global TB Institute Medical Consultants
Meeting, the National TB Conference, the New England TB Clinicians'
Conference, the NYC Annual Genotyping Update, the NYC World TB
Day Medical Conference, the Rutgers Global TB Institute TB Intensive
Workshop, the TB Education and Training Network and TB Program
Evaluation Network Conference and the U.S. Civil Surgeon Symposium.
STAFF PUBLICATIONS IN PEER-REVIEWED JOURNALS, 2019:
1. Hood G, Trieu L, Ahuja SD. Mortality among tuberculosis patients
in New York City. Int J Tuberc Lung Dis. 2019 Feb 1;23(2):252-259.
2. Lam CK, Fluegge K, Macaraig M, Burzynski J. Cost savings
associated with video directly observed therapy for treatment
of tuberculosis. Int J Tuberc Lung Dis. 2019 Nov 1;23(11):
1149-1154.
3. Mase S, Chorba T, Parks S, Belanger A, Dworkin F, Seaworth B,
Warkentin J, Barry P, Shah N. Bedaquiline for the treatment of
multidrug-resistant tuberculosis in the United States. Clin Infect
Dis. 2019 Sep 26. [Epub ahead of print]
4. Nahid P, Mase SR, Migliori GB, Sotgiu G, Bothamley GH, Brozek JL,
Cattamanchi A, Cegielski JP, Chen L, Daley CL, Dalton TL, Duarte
R, Fregonese F, Horsburgh CR Jr, Ahmad Khan F, Kheir F, Lan Z,
Lardizabal A, Lauzardo M, Mangan JM, Marks SM, McKenna L,
Menzies D, Mitnick CD, Nilsen DM, Parvez F, Peloquin CA,
Raftery A, Schaaf HS, Shah NS, Starke JR, Wilson JW, Wortham
JM, Chorba T, Seaworth B. Treatment of drug-resistant
tuberculosis. An ofcial ATS/CDC/ERS/IDSA clinical practice
guideline. Am J Respir Crit Care Med. 2019 Nov 15;200(10):
e93-e142.
5. Narita M, Sullivan Meissner J, Burzynski J. Use of Modeling to
Inform Tuberculosis Elimination Strategies. Am J Respir Crit Care
Med. 2019 Nov 7. [Epub ahead of print]
6. Nguyen MH, Levy NS, Ahuja SD, Trieu L, Proops DC, Achkar JM.
Factors Associated with Sputum Culture-Negative vs
Culture-Positive Diagnosis of Pulmonary Tuberculosis. JAMA
Netw Open. 2019 Feb 1;2(2):e187617.
7. Wilson JW, Nilsen DM, Marks SM. Multidrug Resistant
Tuberculosis in Patients with HIV: Management Considerations
within High-Resourced Settings. Ann Am Thorac Soc. 2019 Jul 31.
[Epub ahead of print]
14 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

>> SPOTLIGHT ON COALITION-BUILDING, EQUITY AND
INTERNAL REFORM, 2019
THE COALITION FOR A TUBERCULOSIS-FREE NYC
THE COALITION FOR A TB-FREE NYC is a collaborative, citywide effort to unite partners across multiple
industries, disciplines, and care levels to develop and implement a strategic plan to eliminate TB
in NYC. Since its inaugural meeting in March 2019, the Coalition has focused on the implications
of the CDC and NTCA’s updated Tuberculosis Screening, Testing, and Treatment of U.S. Health
Care Personnel recommendations and the development of a risk assessment tool for NYC health
care personnel. The Coalition meets quarterly to disseminate information, share resources and
discuss topics of concern to the TB community. All are welcome to join.
>> For more information, please visit the Coalition's website at: tbfreenyc.wixsite.com/tbfreenyc. To join the
Coalition for a TB-Free NYC, please contact [email protected]c.gov.
NYC HEALTH DEPARTMENT RACE TO JUSTICE INITIATIVE
Recognizing that structural racism has been identied as a root cause of gaps and differences in
health outcomes, the Health Department has begun an internal reform initiative, RACE TO JUSTICE
to develop strategies, action plans and the infrastructure needed to advance racial equity and
social justice in policy and practice across all areas of the Agency’s work.
The Health Department aims to promote justice and build
capacity within the Agency to improve health outcomes for
all New Yorkers by:
• Building awareness among staff of how racism
operates within our institutions and structures;
• Developing and implementing strategies to
advance racial equity and social justice across programs, policies and practices;
• Strengthening collaborations with NYC’s communities to counter racist and oppressive
systems within the Health Department and across the city.
To support these aims, the Health Department's Bureau of TB Control participates in the agency's
efforts to plan action steps that will engage all staff in furthering equity and justice in the workplace
and in our interactions with patients and community partners.
In 2019, the Bureau of TB Control conducted open forums in its central ofce and at four Health
Department chest clinics to engage staff in discussions about health and racial equity professional
practices both internally and externally. More than one hundred Bureau staff members participated
in these events, and a clinic-specic workgroup will be launched in 2020.
15 New York City Health Department Annual Tuberculosis Summary, 2019
>> CORE ACTIVITIES

PROFILE OF TB CASES

0
10
20
30
40
50
60
0
500
1000
1500
2000
2500
3000
3500
4000
4500
84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 11 12 13 14 15 16 17 18 19
17 New York City Health Department Annual Tuberculosis Summary, 2019
FIGURE 3: Tuberculosis cases and rates,
1
New York City, 1984-2019
Year
Number of cases
Rate per 100,000
Number of cases
Rate
566 cases
6.9 per 100,0000
There were 566 cases of active TB disease counted in
New York City (NYC) in 2019, an increase of 2% since
2018. The NYC TB incidence rate was 6.9 per 100,000,
more than two times higher than the national TB rate.
1. Rates are based on decennial census data.
2010-2019:
Overall decrease: 20%
Average annual decrease: 2%
1992-2010:
Overall decrease: 81%
Average annual decrease: 8%
1984-1992:
Overall increase: 139%
Average annual increase: 12%

0
1
2
3
4
5
6
7
8
9
10
0
100
200
300
400
500
600
700
800
10 11 12 13 14 15 16 17 18 19
>> PROFILE OF TUBERCULOSIS CASES, 2019
TB IN NEW YORK CITY
Though the number of TB cases in NYC declined by 85% between 1992 and 2019, the rate of decline has
slowed. Since 2010, there has been a 2% average annual decrease in TB cases. Over the past decade, the
overall TB rate has remained relatively stable, with variations in burden among certain populations from
year to year.
FIGURE 4: Tuberculosis cases and rates,
1
New York City, 2010-2019
Year
Number of cases
Rate per 100,000
Number of cases
Rate
1. Rates are based on decennial census data.
FIGURE 5: Percentage point change for select characteristics among patients with tuberculosis disease,
2018-2019, New York City
Female
Male
SEX
+2
-2
0-17
45-64
18-44
65+
-1
+2
-3
+2
BOROUGH OF RESIDENCE
Brooklyn
Queens
Manhattan
The Bronx
+3
-4 -1
+2 +1
Staten
Island
AGE GROUP (YEARS)
RACE/ETHNICITY
1,2
Hispanic
Non-Hispanic
White
Asian
Multiple/
Other
-9
+5
-3
+3
History of
drug use
3
+3
CLINICAL AND SOCIAL CHARACTERISTICS
HIV
infection
+1
-3
HIV status unknown
Culture
positive
+5
History of
health care
work
3,4
+5
1. Race/ethnicity is among patients born in the U.S. 2. There was no change between 2018 and 2019 in the proportion of cases among
U.S.-born Blacks. 3. Within 12 months of diagnosis. 4. Among patients 18 years of age and older.
705 684 652 650 582 575 556 608 553 566
8.6
8.4
8.0
8.0
7.1
7.0
6.8
7.4
6.8
6.9
18 New York City Health Department Annual Tuberculosis Summary, 2019

0
2
4
6
8
10
12
14
16
10 11 12 13 14 15 16 17 18 19
0
50
100
150
200
250
300
350
400
19 New York City Health Department Annual Tuberculosis Summary, 2019
AGE AND SEX
FIGURE 6: Tuberculosis cases by sex,
1
New York
City, 2019
Number of cases
Female Male
FIGURE 7: Tuberculosis cases by age group in
years,
New York City, 2019
40% 60%
1. Data on patient sex are currently collected and
categorized as "Male," "Female" and "Transgender."
In future reports, more expansive categories of gender
identity will be presented to reect changes in data
collection methods.
62%
of TB cases in 2019
occurred among people
who were 45 years of
age or older
1. Rates are based on New York City Health Department population estimates, modied from U.S. Census Bureau interpolated
intercensal population estimates, 2000-2018. Updated August 2019.
FIGURE 8: Tuberculosis rates
1
by age group in years,
New York City, 2010-2019
Year
Rate per 100,000
Age 0-17
Age 45-64Age 18-44 Age 65+
14.0
8.4
5.9
1.1
226
340
11.5
9.4
2.5
10.8
Age 18-44
35%
Age 65+
31%
Age 0-17
3%
Age 45-64
31%
>> PROFILE OF TUBERCULOSIS CASES, 2019

0
10
20
30
40
50
60
70
0
500
1,000
1,500
2,000
2,500
3,000
3,500
92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 11 12 13 14 15 16 17 18 19
0
1
2
3
4
5
6
10 11 12 13 14 15 16 17 18 19
>> PROFILE OF TUBERCULOSIS CASES, 2019
FIGURE 9: Tuberculosis cases and rates
1
by birth in the United States (U.S.),
2,3
New York City, 1992-2019
1. Rates prior to 2000 are based on 1990 U.S. Census data. Rates for 2000-2005 are based on 2000 U.S. Census data. Rates after
2005 are based on one-year American Community Survey data for the given year or the most recent available data. 2. U.S.-born
includes individuals born in the U.S. and U.S. territories. 3. Excludes cases with unknown country of birth.
Year
Number of cases
Rate per 100,000
BIRTH IN THE UNITED STATES
FIGURE 10: Tuberculosis rates
1
by race/ethnicity
2
among patients born in the United States (U.S.),
3,4
New York City, 2010-2019
Year
Rate per 100,000
1. Rates are based on one-year American Community Survey Public Use Microdata Sample data for the given year or the most recent
available data. 2. Data shown do not include patients with multiple, other or unknown race/ethnicity. 3. U.S.-born includes
individuals born in the U.S. and U.S. territories. 4. Excludes cases with unknown country of birth.
Non-Hispanic White
Hispanic
Non-Hispanic Black
Asian
3.4
1.7
0.9
0.7
U.S.-born rate
U.S.-born cases
Non-U.S.-born rate
Non-U.S.-born cases
84%
Proportion of TB cases among
patients born outside the U.S.
71%
Proportion of U.S.-born patients who
were non-Hispanic Black or Hispanic
1.8
91
14.7
474
3,132
59.8
676
32.5
5.7
2.8
1.5
0.9
20 New York City Health Department Annual Tuberculosis Summary, 2019
1. Rates are based on 2018 American
Community Survey one-year sample data.
2. One case in 2019 was in a patient with
unknown country of birth.
3. There were 20 countries for which rate
could not be calculated due to insufcient
population data.
4. China includes individuals born in mainland
China, Hong Kong, Taiwan and Macau.
5. U.S.-born includes individuals born in the
U.S. and U.S. territories.
6. MDR-TB is dened as resistance to at least
isoniazid and rifampin.
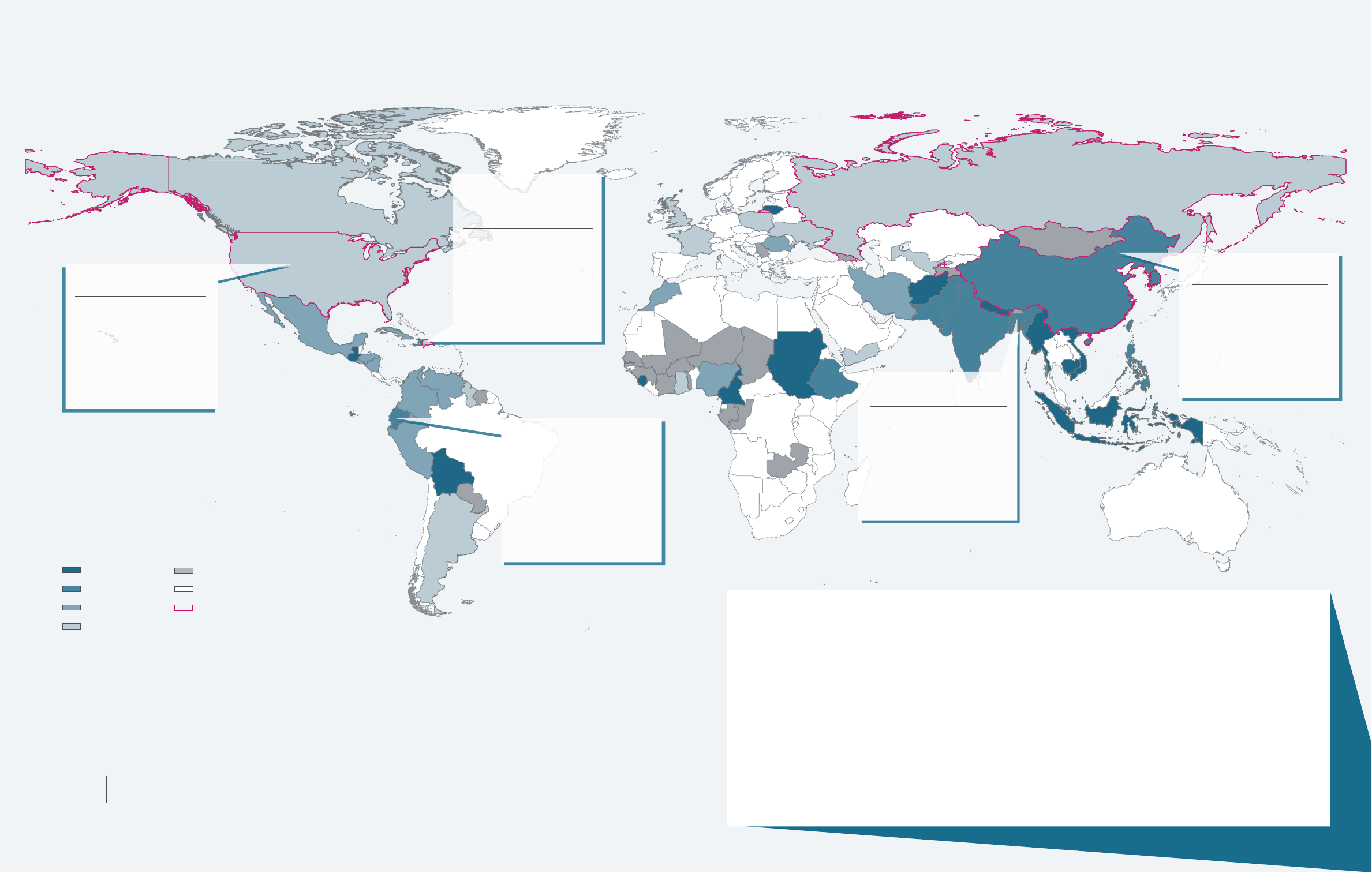
TOP COUNTRIES OF BIRTH BY NEW YORK CITY TB BURDEN AND INCIDENCE,
1,2-3
2019
COUNTRY NUMBER OF CASES
China
4
...............................124
United States (U.S.)
5
........... 91
Ecuador ............................... 31
Dominican Republic ........... 27
Bangladesh ......................... 26
Mexico ................................. 23
India .................................... 20
Philippines .......................... 20
Haiti ..................................... 18
COUNTRY (CASES) NYC TB RATE/100,000
Sudan (1) ............................................ 245.7
Burma (9) ............................................ 149.8
Nepal (14) ........................................... 134.4
Cambodia (1) ...................................... 113.3
Bolivia (3) ............................................ 101.3
Sierra Leone (2) .................................... 97.3
Cameroon (1).........................................72.7
Lithuania (1) ..........................................58.2
Indonesia (2) .........................................56.2
>> PROFILE OF TUBERCULOSIS CASES, 2019
NYC TB rate per 100,000: 26.8
Median age (years): 57
Median years in the U.S.: 8.5
HIV infection: 0%
Diabetes: 54%
26 cases
BANGLADESH
NYC TB rate per 100,000: 31.2
Median age (years): 64
Median years in the U.S.: 16
HIV infection: 0%
Diabetes: 20%
124 cases
CHINA
21 New York City Health Department Annual Tuberculosis Summary, 2019
FIGURE 11: Tuberculosis cases, rates
1
and select characteristics by patient country of birth,
2-5
New York City (NYC), 2019
COUNTRY OF BIRTH
There were 70 countries of birth among patients with TB disease in 2019, and patient characteristics and
TB risk factors differ by country of birth. The Health Department works in partnership with the communities
most affected by TB to develop tailored interventions to reduce TB burden.
70
Number of countries of birth represented
among patients with TB disease in 2019
13
Median number of years in the U.S.
among non-U.S.-born TB patients
NYC TB rate per 100,000: 6.4
Median age (years): 54
Median years in the U.S.: 10
HIV infection: 0%
Diabetes: 26%
27 cases
DOMINICAN
REPUBLIC
NYC TB rate per 100,000: 1.8
Median age (years): 51
Median years in the U.S.: N/A
HIV infection: 16%
Diabetes: 26%
91 cases
UNITED STATES
NYC TB rate per 100,000: 25.6
Median age (years): 46
Median years in the U.S.: 12
HIV infection: 3%
Diabetes: 16%
31 cases
ECUADOR
NYC TB rate per 100,000
40.1 to 245.7
20.1 to 40.0
10.1 to 20.0
1.8 to 10.0
Rate not available
No NYC TB cases
Birth country of at least one
NYC patient with MDR TB
6

FIGURE 12: Tuberculosis rates
1
by United Hospital Fund (UHF) neighborhood, New York City, 2019
TB IN NYC NEIGHBORHOODS
TABLE 1: Proportion of tuberculosis (TB) cases and TB rates
1
by birth in the United States (U.S.)
2-3
and area-based poverty level of patient's residential neighborhood,
4-5
New York City, 2019
1. Rates are based on 2014-2018 American Community Survey data. 2. U.S.-born includes individuals born in the U.S. and U.S.
territories. 3. One case in 2019 had unknown country of birth. 4. Area-based poverty level is based on 2013-2017 American
Community Survey data on the proportion of ZIP code residents living below the federal poverty limit. 5. Cases were assigned to
a ZIP code based on their residence at time of TB diagnosis.
Area-based poverty
level
4-5
U.S.-born
TB rate
% U.S.-born
Non-U.S.-
born rate
% non-
U.S.-born
Total NYC
TB rate
% of
all cases
Very high (30 to 100%) 3.3 32% 15.7 17% 7.8 19%
High (20 to < 30%) 2.3 31% 18.0 30% 8.5 30%
Medium (10 to < 20%) 1.1 24% 14.7 44% 6.9 41%
Low (< 10%) 1.0 13% 7.3 8% 3.0 9%
22 New York City Health Department Annual Tuberculosis Summary, 2019
NYC TB rate per 100,000: 15.0
Most common country of birth
among patients: China (16)
19 cases
SUNSET PARK
NYC TB rate per 100,000: 9.2
Most common country of birth
among patients: United States (5)
13 cases
HUNTS POINT-
MOTT HAVEN
NYC TB rate per 100,000: 19.9
Most common country of birth
among patients: Ecuador (19),
Nepal (12), China (10)
91 cases
WEST QUEENS
At or below provisional national rate (0.7 to 2.7)
Above citywide rate (7.0 to 19.9)
At or below citywide rate (2.8 to 6.9)
Rate per 100,000
>> PROFILE OF TUBERCULOSIS CASES, 2019

6.2
9.2
8.8
8.2
7.2
4.6
2.0
1.1
5.9
15.0
9.7
9.2
5.8
5.6
4.9
4.7
4.4
4.1
3.1
0.7
4.5
10.0
7.8
5.4
4.4
3.8
3.7
3.7
2.4
1.6
0.9
10.0
19.9
18.6
9.2
9.0
7.7
5.7
5.0
4.7
4.2
3.2
4.6
8.7
5.6
4.4
2.6
0.0 2.0 4.0 6.0 8.0 10.0 12.0 14.0 16.0 18.0 20.0
Bronx (89)
Hunts Point-Mott Haven (13)
Crotona-Tremont (19)
Highbridge-Morrisania (18)
Pelham-Throgs Neck (22)
Fordham-Bronx Park (12)
North East Bronx (4)
Kingsbridge-Riverdale (1)
Brooklyn (153)
Sunset Park (19)
Coney Island (28)
East New York (17)
Bedford Stuyvesant-Crown…
Borough Park (19)
Canarsie-Flatlands (10)
East Flatbush-Flatbush (14)
Bensonhurst-Bay Ridge (9)
Williamsburg-Bushwick (9)
Downtown-Brooklyn Heights-…
Greenpoint (1)
Manhattan (74)
Union Square (19)
Central Harlem (14)
Chelsea-Clinton (8)
East Harlem (5)
Gramercy Park-Murray Hill (5)
Washington Heights-Inwood (10)
Upper West Side (8)
Greenwich Village-Soho (2)
Lower Manhattan (1)
Upper East Side (2)
Queens (227)
West Queens (91)
Flushing (47)
Bayside-Little Neck (8)
Fresh Meadows (9)
Southwest Queens (22)
Long Island City-Astoria (12)
Rockaway (6)
Jamaica (15)
Southeast Queens (9)
Ridgewood (8)
Staten Island (22)
Port Richmond (6)
Stapleton-St. George (7)
Willowbrook (4)
South Beach-Tottenville (5)
FIGURE 13: Tuberculosis cases and rates
1,2
by borough and United Hospital Fund (UHF) neighborhood,
New York City, 2019
NOTES (Figure 12, 13):
1. Rates are based on NYC Health Department
population estimates, modied from U.S.
Census Bureau interpolated intercensal
population estimates, 2000-2018.
Updated August 2019.
2. Parentheses indicate the number of TB
cases among patients residing in each
neighborhood at time of TB diagnosis.
Healthy People 2020 goal (1.0 per 100,000)
2019 provisional national rate (2.7 per 100,000)
2019 citywide rate (6.9 per 100,000)
Rate per 100,000
>> PROFILE OF TUBERCULOSIS CASES, 2019
6.2
6.2
9.2
8.8
8.2
7.2
4.6
2.0
1.1
5.9
15.0
9.7
9.2
5.8
5.6
4.9
4.7
4.4
4.1
3.1
0.7
4.5
10.0
7.8
5.4
4.4
3.8
3.7
3.7
2.4
1.6
0.9
10.0
19.9
18.6
9.2
9.0
7.7
5.7
5.0
4.7
4.2
3.2
4.6
8.7
5.6
4.4
2.6
0.0 2.0 4.0 6.0 8.0 10.0 12.0 14.0 16.0 18.0 20.0
The Bronx (89)
Hunts Point-Mott Haven (13)
Crotona-Tremont (19)
Highbridge-Morrisania (18)
Pelham-Throgs Neck (22)
Fordham-Bronx Park (12)
North East Bronx (4)
Kingsbridge-Riverdale (1)
Brooklyn (153)
Sunset Park (19)
Coney Island (28)
East New York (17)
Bedford Stuyvesant-Crown Heights (19)
Borough Park (19)
Canarsie-Flatlands (10)
East Flatbush-Flatbush (14)
Bensonhurst-Bay Ridge (9)
Williamsburg-Bushwick (9)
Downtown-Brooklyn Heights-Park Slope (8)
Greenpoint (1)
Manhattan (74)
Union Square (19)
Central Harlem (14)
Chelsea-Clinton (8)
East Harlem (5)
Gramercy Park-Murray Hill (5)
Washington Heights-Inwood (10)
Upper West Side (8)
Greenwich Village-Soho (2)
Lower Manhattan (1)
Upper East Side (2)
Queens (227)
West Queens (91)
Flushing (47)
Bayside-Little Neck (8)
Fresh Meadows (9)
Southwest Queens (22)
Long Island City-Astoria (12)
Rockaway (6)
Jamaica (15)
Southeast Queens (9)
Ridgewood (8)
Staten Island (22)
Port Richmond (6)
Stapleton-St. George (7)
Willowbrook (4)
South Beach-Tottenville (5)
6.2
9.2
8.8
8.2
7.2
4.6
2.0
1.1
5.9
15.0
9.7
9.2
5.8
5.6
4.9
4.7
4.4
4.1
3.1
0.7
4.5
10.0
7.8
5.4
4.4
3.8
3.7
3.7
2.4
1.6
0.9
10.0
19.9
18.6
9.2
9.0
7.7
5.7
5.0
4.7
4.2
3.2
4.6
8.7
5.6
4.4
2.6
0.0 2.0 4.0 6.0 8.0 10.0 12.0 14.0 16.0 18.0 20.0
Bronx (89)
Hunts Point-Mott Haven (13)
Crotona-Tremont (19)
Highbridge-Morrisania (18)
Pelham-Throgs Neck (22)
Fordham-Bronx Park (12)
North East Bronx (4)
Kingsbridge-Riverdale (1)
Brooklyn (153)
Sunset Park (19)
Coney Island (28)
East New York (17)
Bedford Stuyvesant-Crown…
Borough Park (19)
Canarsie-Flatlands (10)
East Flatbush-Flatbush (14)
Bensonhurst-Bay Ridge (9)
Williamsburg-Bushwick (9)
Downtown-Brooklyn Heights-…
Greenpoint (1)
Manhattan (74)
Union Square (19)
Central Harlem (14)
Chelsea-Clinton (8)
East Harlem (5)
Gramercy Park-Murray Hill (5)
Washington Heights-Inwood (10)
Upper West Side (8)
Greenwich Village-Soho (2)
Lower Manhattan (1)
Upper East Side (2)
Queens (227)
West Queens (91)
Flushing (47)
Bayside-Little Neck (8)
Fresh Meadows (9)
Southwest Queens (22)
Long Island City-Astoria (12)
Rockaway (6)
Jamaica (15)
Southeast Queens (9)
Ridgewood (8)
Staten Island (22)
Port Richmond (6)
Stapleton-St. George (7)
Willowbrook (4)
South Beach-Tottenville (5)
23 New York City Health Department Annual Tuberculosis Summary, 2019

0
50
100
150
200
250
300
350
400
450
500
92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 11 12 13 14 15 16 17 18 19
FIGURE 14: Multidrug resistance
1
among tuberculosis cases, New York City (NYC), 1992-2019
1. MDR-TB is dened as resistance to at least isoniazid and rifampin. 2. XDR-TB is dened as resistance to at least isoniazid and rifampin
plus a uoroquinolone and a second-line injectable anti-TB medication.
Multidrug-resistant TB (MDR-TB)
1
Extensively drug-resistant
TB (XDR-TB)
2
INSET: MDR-
1
and XDR-TB
2
in NYC,
2010-2019
Year
Number of cases
Number of cases
Year
DRUG RESISTANCE
In 2019, 11 patients were newly-diagnosed with MDR-TB, dened as a TB strain resistant to both isoniazid and
rifampin, two of the most effective TB drugs. Molecular-based tests, which can rapidly detect mutations
associated with drug resistance, are now being used routinely in hospitals, commercial laboratories and public
health reference laboratories.
Characteristics
Median age (range) 57 (32-84)
Number born outside of the United States (%) 10 (91%)
Pulmonary site of disease (%) 11 (100%)
Median number of drugs to which there was
known resistance among MDR-TB cases
2
(range)
7 (3-10)
Median number of contacts identied around
patients with MDR-TB (range)
3 (0-55)
0
5
10
15
20
10 11 12 13 14 15 16 17 18 19
REGION OF BIRTH
3
1. MDR-TB is dened as resistance to at least isoniazid and rifampin.
2. Resistance to any uoroquinolone was counted once.
3. Based on World Health Organization regional denitions.
TABLE 2: Select characteristics among patients diagnosed with multidrug-resistant tuberculosis (MDR-TB),
1
New York City, 2019 (n=11)
24 New York City Health Department Annual Tuberculosis Summary, 2019
11
16
18
7
10
5
11
15
13
11
437
289
183
114
84
57
38
31
24
25
29
22
19
24
24
9
11
9
54%
of patients with
MDR-TB were born
in Europe or the
Americas
Western Pacic
37%
Americas
27%
South-East Asia
9%
Europe
27%
11
16
18
7
10
5
11
15
13 11
>> PROFILE OF TUBERCULOSIS CASES, 2019

TABLE 4: Detection of resistance to select tuberculosis medications by test type,
1
New York City, 2019
INH RIF EMB PZA FLQ
2
INJ
3
Number of patients with a molecular
test conducted
4
418 450 403 403 411 403
>> Number with mutation detected (%) 36 (9%) 15 (3%) 3 (1%) 15 (4%) 5 (1%) 3 (1%)
Number of patients with a phenotypic
test conducted
397 395 392 390 67 61
>> Number with resistance detected (%) 46 (12%) 12 (3%) 8 (2%) 20 (5%) 5 (7%) 4 (7%)
Number of patients with any drug
susceptibility test conducted
440 461 425 427 412 404
>> Number with resistance detected (%) 47 (11%) 15 (3%) 8 (2%) 24 (6%) 6 (1%) 4 (1%)
1. Categories are not mutually exclusive. 2. Fluoroquinolones include levooxacin, moxioxacin, ciprooxacin and ooxacin. 3. Injectables
include kanamycin, capreomycin and amikacin. 4. Molecular tests include GeneXpert
®
MTB/RIF, Genotype MTBDRplus (Hain),
pyrosequencing, Sanger sequencing and whole genome sequencing.
TABLE 3: Number of tests performed, gene targets and median turnaround time for select molecular tests
for tuberculosis, New York City, 2019
1. Fluoroquinolones include levooxacin, moxioxacin, ciprooxacin and ooxacin. 2. Injectables include kanamycin, capreomycin and
amikacin. 3. Turnaround time is dened as the number of days between the earliest date a result is reported and the collection date of
that respective specimen.
Test type
Gene targets and corresponding medication
Number
of patients
with test
conducted
Median
turnaround
time (days)
3
INH RIF EMB PZA FLQ
1
INJ
2
• inhA
• katG
• oxyR-ahpC PR
• mabA-inhA PR
• mabA
• rpoB
• embB
• embC-
embA PR
• pncA
• pncA PR
• gyrA
• gyrB
• rrs1400
• eis
• tlyA
GeneXpert
®
MTB/RIF
•
229 2
Pyrosequencing
• • •
89 15
Whole genome
sequencing
• • • • • • •
403 49
11
Number of patients newly-diagnosed
with an MDR-TB strain in 2019
97%
Percent decrease in the number of
MDR-TB cases in NYC from 1992-2019
13%
Proportion of cases with resistance
to at least one rst-line drug
30
Number of patients with MDR-TB treated at
a Health Department chest clinic in 2019
>> PROFILE OF TUBERCULOSIS CASES, 2019
Abbreviations: INH - Isoniazid; RIF - Rifampin; EMB - ethambutol; PZA - pyrazinamide; FLQ - uoroquinolones; INJ - injectables
Abbreviations: INH - Isoniazid; RIF - Rifampin; EMB - ethambutol; PZA - pyrazinamide; FLQ - uoroquinolones; INJ - injectables
25 New York City Health Department Annual Tuberculosis Summary, 2019

1. U.S.-born includes individuals born in the U.S. and U.S. territories. 2. Excludes cases with unknown country of birth. 3. There
were 87 patients in 2019 with an unknown HIV status. 4. Immunosuppression due to use of immunosuppressive therapy or
having a medical condition, not including HIV/AIDS.
DISEASE SITE, CULTURE STATUS AND CO-MORBIDITIES
FIGURE 15: Tuberculosis cases by disease site,
New York City, 2019
1. Categories are not mutually exclusive.
FIGURE 16: Proportion of culture-conrmed tuberculosis cases among all cases, New York City, 2019
TABLE 5: Disease site among tuberculosis cases with
extrapulmonary disease,
1
New York City, 2019 (n=191)
Disease site
Number
of cases
Percent
Any extrapulmonary site 191 -
Lymphatic 80 42%
Pleural 61 32%
Bone/joint 20 10%
Peritoneal 11 6%
Genitourinary 9 5%
Meningeal 4 2%
Laryngeal 1 1%
Other 36 19%
81%
of all cases
had pulmonary
involvement
FIGURE 17: Select co-morbidities among patients with tuberculosis disease by birth in the United States
(U.S.),
1,2
New York City , 2015-2019
HIV INFECTION
3
Proportion of cases
Year
Proportion of cases
Year
Proportion of cases
Year
DIABETES IMMUNOSUPRESSION (NON-HIV)
4
Culture-positive 82%
Case conrmed without positive culture 18%
U.S.-born
Non-U.S.-born
Overall proportion, 2019: 6% Overall proportion, 2019: 23% Overall proportion, 2019: 7%
0
5
10
15
20
25
30
15 16 17 18 19
0
5
10
15
20
25
30
15 16 17 18 19
0
5
10
15
20
25
30
15 16 17 18 19
Both pulmonary and
extrapulmonary disease sites
15%
Pulmonary
disease only
66%
Extrapulmonary disease only
19%
9
16
10
12
17
4
3
5
3
4
26
23
18
11
12
13
21
20 20
24
6 6 6
8
7
6
9
7
7
5
>> PROFILE OF TUBERCULOSIS CASES, 2019
26 New York City Health Department Annual Tuberculosis Summary, 2019

0
2
4
6
8
10
0
10
20
30
40
50
60
14 15 16 17 18
MORTALITY, TREATMENT COMPLETION AND INDICATORS
FIGURE 19: Number and proportion of patients
with tuberculosis (TB) who died
1,2
before or during
treatment, New York City (NYC), 2014-2018
1. Treatment outcomes are not reported for the current year to allow sufcient time for follow-up. 2. A death is dened as any
patient who died prior to or during TB treatment, regardless of the cause of death. This excludes any patient who died after
the completion of TB treatment.
Died prior to TB treatment completion
Died prior to TB treatment initiation
Proportion of all NYC TB patients who died
FIGURE 18: Treatment outcomes for tuberculosis (TB)
cases counted in 2018,
1,2
New York City (n=553)
TABLE 6: Select performance measures, national targets
1
and New York City outcomes, 2017-2018
Indicator 2017 2018 2020 target
Treatment and case management for persons with active tuberculosis (TB)
Initiated TB treatment within seven days of specimen collection
2
92% 88% 97%
Sputum culture conversion within 60 days of treatment initiation
3
67% 74 % 73%
Completed treatment within 365 days of initiation
4
92% 92% 95%
Contact investigation
Eligible cases with contacts elicited
5
95% 93% 100%
Eligible contacts evaluated
6
85% 83% 93%
Eligible contacts who initiated treatment for TB infection
7
85% 86% 91%
Eligible contacts who completed treatment for TB infection
8
82% 62% 81%
1. Denitions for performance measures and national indicators are established by the CDC; the 2020 targets were set in 2015.
For details, visit cdc.gov/tb/programs/evaluation/indicators. Performance measures are not reported for the current year to allow
sufcient time for follow-up. 2. Of TB patients with positive acid-fast bacilli (AFB) sputum-smear results who were alive at diagnosis.
3. Of TB patients with positive sputum culture results who were alive at diagnosis and have initiated treatment. Excludes patients who
died within 60 days of initiating treatment. 4. Excludes patients who never started on anti-TB medications, those who died or moved
outside of the U.S. within 365 days of treatment initiation, those with any rifampin resistance, those with meningeal TB and children
14 years of age or younger with disseminated TB. 5. Of AFB sputum smear-positive TB patients. 6. Of contacts to AFB sputum smear-
positive TB patients counted in the year of interest. 7. Of contacts to AFB sputum smear-positive TB patients who have newly diagnosed
TB infection. 8. Of contacts to sputum AFB smear-positive TB patients with newly diagnosed TB infection who started treatment.
>> PROFILE OF TUBERCULOSIS CASES, 2019
Number of cases
Year
Proportion of cases
82%
of all patients with
TB disease in
2018 completed
treatment
Lost to follow-up 2%
Refused treatment 2%
Moved outside U.S. 1%
Currently on treatment
5%
Died 9%
Completed
treatment
82%
37
12
28
11
25
14
32
13
35
13
8
7 7
7
9
27

FIGURE 20: Contact investigations in non-household
settings
1
by site type, New York City, 2019 (n=59)
CONTACT INVESTIGATION IN NON-HOUSEHOLD SETTINGS
The Health Department investigates TB exposures in non-household settings in NYC to identify and evaluate
contacts, ensure appropriate treatment for contacts with TB disease or LTBI, determine if transmission has
occurred and assess whether testing of additional contacts may be warranted.
1. Excludes health care-associated investigations. 2. Proportion calculated among investigations where transmission could be
assessed. 3. Contacts eligible for testing are dened as contacts without a known history of TB disease or documented positive test
for TB infection who were alive after diagnosis of the infectious TB case to whom they were exposed.
TABLE 7: Contact investigation outcomes in non-household settings
1
by number of exposed contacts,
New York City, 2019 (n=59)
≥ 15 exposed contacts < 15 exposed contacts Total
n (%) n (%) n (%)
Number of sites 20 34 39 66 59
Likely transmission
2
7 39 7 21 14 27
Transmission could not be assessed 2 10 5 13 7 12
Total number of contacts 592 - 270 - 862 -
Median contacts per site (range) 26 (12-66) 6 (1-38) 9 (1-66)
Contacts eligible for testing
3
575 97 257 95 832 97
Contacts tested 511 89 240 93 751 90
Contacts with a positive TB test result 43 8 29 12 72 10
FIGURE 21: Contact investigations in health
care-associated settings by site type, New York City,
2019 (n=216)
78%
of contact investigations
in non-household settings
were conducted
in workplaces
216
health care-associated
settings were investigated
around exposures for
157 patients
28 New York City Health Department Annual Tuberculosis Summary, 2019
Workplace (78%)
Adult day center (3%) Place of worship (3%)
Homeless shelter/Residential facility (3%)
School/Day care (5%)Other (7%)
Acute care facility
1
(59%)
Home health care agency (25%)
Nursing home/long-term care facility (7%)
Other outpatient health care facility (7%)
Other facilities (1%)
1. Includes hospitals and acute care clinics
78%
7%
5%
3%
3%
3%
59%
25%
7%
7%
1%
>> PROFILE OF TUBERCULOSIS CASES, 2019

WHOLE GENOME SEQUENCING (WGS) AND NYC CLUSTERS
The Health Department uses WGS to characterize TB strains and assess transmission. Universal WGS has
been performed for culture-positive cases since 2016. Cases whose isolates are within 20 single nucleotide
polymorphisms (SNP) of another case in NYC or NYS are notied to NYC by NYS Wadsworth laboratory. Notied
cases are reviewed, prioritized and assigned for epidemiologic investigation. A difference of ve SNPs or less
between isolates is considered suggestive of possible recent transmission and prompts further investigation.
1. Clusters include cases whose isolates have less than or equal to ve single nucleotide polymorphism differences among them
based on whole genome sequencing results, as well as clinically-counted cases with conrmed epidemiologic links to cluster cases.
2. Includes clusters with three or more cases identied in three years and evidence of recent, local TB transmission. 3. Includes one
case counted outside of NYC. 4. Among cluster cases identied between January 1, 2017 and December 31, 2019. 5. U.S.-born
includes individuals born in the U.S. and U.S. territories. 6. In the 12 months before TB diagnosis. 7. Within a 10-block radius or less.
TABLE 8: Characteristics of select high-priority whole genome sequencing-identied tuberculosis (TB)
clusters,
1,2
New York City, 2019
Cluster A
3
Cluster B Cluster C Cluster D Cluster E
Number of cases identied between
Jan. 1, 2017 and Dec. 31, 2019
6 6 4 4 4
Proportion of cases among males
4
33% 100% 75% 50% 25%
Proportion of patients born in the
United States (U.S.)
4,5
100% 0% 0% 50% 100%
Median patient age in years (range)
4
39 (19-62) 40 (22-50) 34 (29-38) 49 (25-62) 24 (2-58)
Most common borough of residence at
time of TB diagnosis (%)
4
Manhattan (33%)
The Bronx (33%)
Manhattan
(67%)
Queens
(50%)
Brooklyn
(100%)
The Bronx
(100%)
Proportion of patients reporting history
of homelessness
4,6
0% 83% 0% 0% 50%
Proportion of patients reporting history
of drug use or excessive alcohol use
4,6
83% 83% 50% 50% 25%
Clusters in which patients reported
history of transient work
4,6
• •
Clusters in which social network links
were identied among patients
4
• • •
Clusters in which patients had links
to the same neighborhood
4,7
• •
>> PROFILE OF TUBERCULOSIS CASES, 2019
29
FIGURE 22: Tuberculosis cases by whole genome sequencing (WGS) availability and high quality single
nucleotide polymorphism (SNP) analysis results, New York City, 2019 (n=566)
>20 SNPs from any other TB case
1
62%
Culture positive, WGS not available 4%
0-5 SNPs from at least one other TB case
1
7%
6-20 SNPs from at least one other TB case
1
9%
Culture negative 18%
1. May include cases veried outside of New York City with WGS
results in the NYS Wadsworth laboratory database.

Characteristics
2018 2019
U.S.-born
1
Non-U.S.-born Total
2
U.S.-born
1
Non-U.S.-born Total
2
n % n % n % n % n % n %
Age group
0-17 12 14% 11 2% 23 4% 12 13% 7 1% 19 3%
18-44 27 32% 183 39% 211 38% 21 23% 179 38% 200 35%
45-64 23 27% 135 29% 158 29% 34 37% 138 29% 173 31%
65+ 23 27% 138 30% 161 29% 24 26% 150 32% 174 31%
Sex
Female 30 35% 182 39% 212 38% 39 43% 186 39% 226 40%
Male 55 65% 285 61% 341 62% 52 57% 288 61% 340 60%
Race/ethnicity
Non-Hispanic White 9 11% 26 6% 35 6% 15 16% 23 5% 38 7%
Non-Hispanic Black 38 45% 58 12% 96 17% 41 45% 61 13% 103 18%
Hispanic 30 35% 105 22% 136 25% 24 26% 124 26% 148 26%
Asian 5 6% 256 55% 261 47% 3 3% 232 49% 235 42%
Multiple/other 3 4% 22 5% 25 5% 6 7% 34 7% 40 7%
Time in the U.S. (at time of reporting)
< 1 year n/a n/a 25 5% 25 5% n/a n/a 24 5% 24 5%
1-5 years n/a n/a 112 24% 112 24% n/a n/a 123 26% 123 26%
> 5 years n/a n/a 328 70% 328 70% n/a n/a 322 68% 322 68%
Borough of residence 14 17 225 47 239 43 16 19 231 44 247 40
Manhattan 21 25% 58 12% 79 14% 16 18% 58 12% 74 13%
The Bronx 24 28% 56 12% 80 14% 22 24% 67 14% 89 16%
Brooklyn 28 33% 144 31% 173 31% 28 31% 124 26% 153 27%
Queens 9 11% 193 41% 202 37% 20 22% 207 44% 227 40%
Staten Island 3 4% 16 3% 19 3% 5 5% 17 4% 22 4%
Neighborhood poverty
3
Low (< 10%) 9 11% 44 9% 53 10% 12 13% 38 8% 50 9%
Medium (10 to < 20%) 17 20% 212 45% 229 41% 22 24% 209 44% 232 41%
High (20 to < 30%) 28 33% 128 27% 156 28% 28 31% 144 30% 172 30%
Very High (30 to 100 %) 29 34% 78 17% 107 19% 29 32% 80 17% 109 19%
Homeless
4
7 8% 13 3% 20 4% 6 7% 13 3% 19 3%
Employed
4,5
28 38% 187 41% 215 41% 32 41% 195 42% 227 41%
Health care worker
4,5
1 4% 14 7% 15 7% 2 6% 25 13% 27 12%
Drug use
4
16 19% 7 1% 23 4% 19 21% 17 4% 37 7%
Excessive alcohol use
4
6 7% 15 3% 22 4% 4 4% 19 4% 23 4%
TABLE 9: Select demographic, social and clinical characteristics of tuberculosis cases by birth in the
United States (U.S.),
1
New York City, 2018-2019
30 New York City Health Department Annual Tuberculosis Summary, 2019
>> PROFILE OF TUBERCULOSIS CASES, 2019

Characteristics
2018 2019
U.S.-born
1
Non-U.S.-born Total
2
U.S.-born
1
Non-U.S.-born Total
2
n % n % n % n % n % n %
Ever respiratory smear
positive
6
41 59% 201 54% 243 55% 44 59% 225 59% 269 59%
Sputum smear positive
6
39 95% 186 93% 225 93% 42 95% 211 94% 253 94%
Culture positive 58 68% 362 78% 421 77% 77 85% 385 82% 463 82%
Pulmonary only site of
disease
60 71% 294 63% 355 64% 62 68% 312 66% 375 66%
Extrapulmonary only site
of disease
16 19% 94 20% 110 20% 16 18% 90 19% 106 19%
Both pulmonary and
extrapulmonary sites
9 11% 79 17% 88 16% 13 14% 72 15% 85 15%
Cavities present on chest
x-ray ever
6
23 33% 70 19% 93 21% 20 27% 66 17% 86 19%
Multidrug (MDR) resistance
7
2 3% 11 3% 13 3% 1 1% 10 3% 11 3%
Extensive drug resistance
(XDR)
8
0 0 0 0 0 0 0 0% 0 0 0 0%
Non-MDR isoniazid
resistance
9
7 12% 33 9% 40 9% 4 5% 32 9% 36 8%
Non-MDR rifampin
resistance
10
0 0 2 1% 2 0 1 1% 3 1% 4 1%
History of TB disease 3 4% 40 9% 43 8% 4 4% 39 8% 43 8%
HIV status
Infected 10 12% 15 3% 25 5% 15 16% 20 4% 36 6%
Not infected 52 61% 377 81% 430 78% 60 66% 383 81% 443 78%
Refused testing 18 21% 59 13% 77 14% 11 12% 42 9% 53 9%
Not offered/done or
unknown
5 6% 16 3% 21 4% 5 5% 29 6% 34 6%
Non-HIV related
immunosuppression
8 9% 34 7% 42 8% 5 5% 35 7% 40 7%
Diabetes 15 18% 112 24% 127 23% 24 26% 108 23% 133 23%
Total 85 15 467 84 553 - 91 16 474 84 566 -
1. U.S.-born includes individuals born in the U.S. and U.S. territories. 2. Column sums may not equal applicable totals due to
missing country of birth data. 3. Area-based poverty level is based on 2013-2017 American Community Survey data on the
proportion of ZIP code residents living below the federal poverty level. Cases were assigned to a ZIP code based on their residence
at TB diagnosis. 4. In the 12 months before TB diagnosis. 5. Among patients 18 years of age and older. 6. Percent is among
patients with a pulmonary site of disease. 7. MDR-TB is dened as resistance to at least isoniazid and rifampin. Percent is among
patients with susceptibility testing performed for isoniazid and rifampin. 8. XDR-TB is dened as resistance to at least isoniazid and
rifampin plus a uoroquinolone and a second-line injectable anti-TB medication. Percent is among patients with susceptibility testing
performed for isoniazid, rifampin, any uoroquinolone and any second-line injectable anti-TB medication. 9. Percent is among patients
with susceptibility testing performed for isoniazid who did not have MDR-TB. 10. Percent is among patients with susceptibility testing
performed for rifampin who did not have MDR-TB.
>> PROFILE OF TUBERCULOSIS CASES, 2019
31 New York City Health Department Annual Tuberculosis Summary, 2019

APPENDICES

TB REPORTING REQUIREMENTS
Medical, dental, osteopathic and other health care providers and administrators of hospitals or other institutions providing
care and treatment, or their designees, including infection control practitioners, are required by the NYC Health Code
§§11.03 and 11.05 to report all patients, alive or deceased, with suspected or conrmed TB disease to the NYC Health
Department within 24 hours of diagnosis or clinical suspicion. Medical providers must report these patients even though
microbiologists and pathologists are also required to report ndings consistent with TB. Reports must be submitted
using the Universal Reporting Form (URF) and must be received by the Health Department within 24 hours of diagnosis or
clinical suspicion, whether sent electronically, by express or overnight mail, fax or telephone.
HEALTH CARE PROVIDERS
Health care providers in NYC are encouraged to submit reports electronically through a NYCMED account. Alternatively,
providers may fax a completed URF to the Health Department's Bureau of TB Control at 844-713-0557. Information reported on
the URF should be as complete as possible. The following essential information must be included when the report is submitted
to the Health Department:
• Information needed to identify and locate the individual (e.g., name, telephone, address, date of birth)
• Provider information (e.g., physician’s name, reporting facility, phone number, email)
• Results of acid-fast bacilli (AFB) smear, including specimen source, date specimen obtained and accession number
• Results of radiologic exams (e.g., X-ray or imaging)
• Any treatment information
• Quantitative and qualitative results from TST or blood-based IGRA test for children younger than 5 years of age who have
a positive test for TB infection
MICROBIOLOGY AND PATHOLOGY LABORATORIES
Laboratories are required to report via the NYS Electronic Clinical Laboratory Reporting System (ECLRS). Per the NYC Health
Code sections §§13.03 and 13.05, the following results must be reported to the Health Department, whether conrmed or
presumptive, for patients alive or deceased, within 24 hours of obtaining test results:
• AFB-positive smears (regardless of anatomic site)
• Nucleic acid amplication (NAA) test results and cultures positive for M. tuberculosis complex
• Results of susceptibility tests performed on M. tuberculosis complex cultures
• Biopsy, pathology or autopsy ndings consistent with TB disease, including but not limited to presence of AFB on smear
and caseating and/or necrotizing granulomas that are consistent with TB in the lung, lymph nodes or other specimens
• Any culture or NAA result associated with an AFB-positive smear (even if negative for M. tuberculosis complex)
• For patients with a positive TB diagnostic laboratory result, all subsequent TB diagnostic laboratory results (negative or
positive) from specimens collected within one year of the most recent positive result
• Quantitative and qualitative results from blood-based IGRA tests regardless of test result or patient age
>> Health Code §13.05(a) also mandates that a portion of the initial culture be sent for DNA analysis to the NYC Public
Health Laboratory (455 First Ave., Room 236; New York, NY 10016) within 24 hours of observing growth of
M. tuberculosis complex in a culture from any specimen. A specimen submitted to the Health Department for drug
susceptibility testing meets this requirement unless the Health Department noties the submitter otherwise.
► Reporting should never be delayed pending identication of M. tuberculosis with an NAA test or culture.
Patients should be reported whenever TB is suspected, even if bacteriologic evidence of disease is lacking
or treatment has not been initiated. If TB treatment is initiated after submitting the initial disease report,
the provider is required to submit an updated report.
33 New York City Health Department Annual Tuberculosis Summary, 2019
>> REPORTING REQUIREMENTS

REPORTING PATIENT FOLLOW-UP
Health Code §11.21(a)(3) requires the treating physician to report whether the patient completed treatment and the patient’s
treatment outcome (i.e., cured, failed, relapsed, lost, moved, refused), or whether treatment was discontinued if the patient
was found not to have TB or for another reason. Physicians must assist the Health Department with evaluation and follow-up for
persons suspected of having TB. Case managers contact the treating physicians to request updates and ensure that appropriate
treatment and monitoring are being conducted. Health care providers must provide access to necessary paper and electronic
medical records to authorized Health Department staff as requested [Health Code §11.03(e)].
Additionally, Health Code §11.21(a)(1) requires that the treating physicians or persons in charge of facilities must submit
monthly clinical status reports for patients with TB disease, which must include at least:
• Name, address and telephone number(s) of the patient
• Whether treatment is still ongoing
• The clinical status and treatment being provided
• Dates and results of sputum and X-ray exams
• Any other information required by the Health Department
To facilitate mandatory monthly patient status reports, the Health Department created the “Report of Patient Services” form
(TB 65). This form, or a report containing the same information, must be submitted to the patient’s case manager.
When requested by the Health Department, medical providers are also required to report all information on the evaluation,
testing and treatment of individuals who have been in contact with a person with TB disease. [Health Code §11.21(b)]
SUBMITTING HOSPITAL DISCHARGE AND TB TREATMENT PLANS
Health Code §11.21(a)(4) requires health care providers to submit a discharge plan to the Health Department for review and
approval prior to discharging infectious TB patients from the hospital. The Hospital Discharge Approval Request Form (TB 354)
must be submitted 72 hours before the planned discharge date and must be approved by the Health Department prior to
discharge.
For more information and to download related forms, call 311 and ask for the BTBC Surveillance Unit or visit
nyc.gov and search for TB reporting requirements. To download a URF, search for URF. To create a NYCMED account,
search for NYCMED. To download forms related to hospital discharge plans and reporting patient services, search
for TB reporting requirements.
IT IS MANDATORY TO REPORT PATIENTS WHO MEET ANY OF THE FOLLOWING CRITERIA:
• Positive NAA test result (e.g., Gen-Probe® Amplied™
Mycobacterium Tuberculosis [MTD] test, Cepheid
GeneXPert® MTB/RIF, Hain Lifescience GenoType
MTBDRplus, Hain Lifescience GenoType MTBDRsl)
for M. tuberculosis complex
• Positive culture for M. tuberculosis complex,
including: M. tuberculosis, M. africanum, M. bovis-
BCG, M. caprae, M. canetti, M. microti, M. pinnipedii,
M. bovis, M. dassie, M. mungi, M. orygis
• Biopsy, pathology or autopsy ndings consistent
with TB disease, including caseating or necrotizing
granulomas in biopsy of lung, lymph nodes or
other specimens
• Quantitative and qualitative results from blood-based
IGRA tests regardless of test result or patient age
• Positive smear (from any anatomical site) for AFB
• Clinical suspicion of pulmonary or extrapulmonary
TB such that the health care provider has initiated
or intends to initiate isolation or treatment for TB
disease with two or more anti-TB medications
• Any child younger than 5 years of age (on the day
of test administration up to the day of the fth
birthday), with a positive TST or IGRA result,
regardless of whether the child has received a
Bacille Calmette-Guerin (BCG) vaccination. For
these patients, providers must report quantitative
and qualitative results from blood-based IGRA tests
or induration (millimeters) for TSTs, as well as related
chest imaging results and any preventive medication
initiated for LTBI.
>> REPORTING REQUIREMENTS
34 New York City Health Department Annual Tuberculosis Summary, 2019

TB REPORTING DATA
FIGURE 24: Children younger than 5 years of age
reported to the Health Department with a positive
test result for tuberculosis infection
1
by reporter
type, New York City, 2015-2019
Number of cases
Year
Laboratory
2
Health care provider
Patient characteristics
Median patient age (range) 2 (0-4)
Received a QuantiFERON® test (%)
2
60 (55%)
Received a tuberculin skin test (%)
2
51 (47%)
Initiated treatment (%) 57 (52%)
TABLE 12: Select characteristics of children younger
than 5 years of age with a positive test for
tuberculosis infection
1
result reported to the
New York City Health Department, 2019 (n=109)
1. Includes tuberculin skin tests, QuantiFERON®-TB Gold tests
and T-SPOT.TB tests. 2. Test type categories are not mutually
exclusive.
FIGURE 23: Initial reporter of conrmed
tuberculosis cases veried in New York
City by reporter type, 2019
TABLE 11: Most common non-laboratory reporting facilities
by number of conrmed tuberculosis cases reported, New
York City, 2019
Facility
Number
of cases
NYC Health+Hospitals/Elmhurst 46
New York-Presbyterian Queens 35
Maimonides Medical Center 25
New York-Presbyterian/Weill Cornell Medical Center 21
Mount Sinai Hospital 19
New York-Presbyterian/Brooklyn Methodist Hospital 18
Monteore Medical Center - Henry and Lucy
Moses Division
16
NYC Health+Hospitals/Lincoln 14
NYC Health+Hospitals/Queens 14
Long Island Jewish Medical Center 13
NYC Health+Hospitals/Bellevue 13
New York-Presbyterian/Columbia University
Medical Center
13
Non-public hospital (55%)
Public hospital (24%)
Community provider (8%)
Health Department chest clinic (5%)
Commercial laboratory (5%)
Non-NYC reporter (3%)
Other (<1%)
78%
of TB cases were
rst reported by
a hospital
0
20
40
60
80
100
120
15 16 17 18 19
1. Includes tuberculin skin tests, QuantiFERON®-TB Gold tests
and T-SPOT.TB tests. 2. Laboratory reporting was added to
the Health Code in 2017.
109
97
71
66
90
55%
24%
8%
5%
5%
3%
1%
>> TUBERCULOSIS REPORTING DATA
35 New York City Health Department Annual Tuberculosis Summary, 2019

TABLE 10: Tuberculosis (TB) cases and rates
1
by select characteristics, New York City, 1900-2019
1. Rates are based on decennial census data. 2. Multidrug-resistant TB (MDR-TB) is dened as resistance to at least isoniazid and rifampin. 3. Data on TB deaths are obtained from the
Health Department's Ofce of Vital Statistics. Deaths recorded in a given year may include cases diagnosed in a previous year.
Year
Number of
TB cases
Rate per
100,000
Cases
with positive
culture
Cases with
positive
sputum smear
Sputum smear
positive rate
per 100,000
Multidrug-
resistant
cases
2
Deaths
attributable
to TB
3
Death rate
per 100,000
1900 11,997 349.0 9,630 280.2
1910 32,065 672.7 10,074 211.3
1920 14,035 249.7 7,915 140.8
1930 11,821 170.6 4,574 66.0
1940 9,005 120.8 3,680 49.4
1950 7,717 97. 8 2,173 27.5
1960 4,699 60.4 824 10.6
1970 2,590 32.8 432 5.5
1980 1,514 21.4 143 2.0
1981 1,582 22.4 155 2.2
1982 1,583 22.4 168 2.4
1983 1,603 22.7 151 2.1
1984 1,573 22.2 1,485 168 2.4
1985 1,811 25.6 1,756 155 2.2
1986 2,197 31.1 2,156 186 2.6
1987 2,166 30.6 2,129 219 3.1
1988 2,281 32.3 2,205 246 3.5
1989 2,535 35.8 2,404 236 3.3
1990 3,506 47.9 3,384 256 3.5
1991 3,653 49.9 3,462 1,826 24.9 385 245 3.3
1992 3,755 51.3 3,401 1,855 25.3 437 200 2.7
1993 3,151 43.0 2,784 1,529 20.9 289 166 2.3
1994 2,941 40.2 2,433 1,280 17.5 183 133 1.8
1995 2,408 32.9 1,996 1,001 13.7 114 94 1.3
1996 2,013 27.5 1,693 873 11.9 84 67 0.9
1997 1,705 23.3 1,383 708 9.7 57 55 0.8
1998 1,528 20.9 1,232 611 8.3 38 52 0.7
1999 1,436 19.6 1,124 571 7.8 31 49 0.7
2000 1,311 16.4 1,043 516 6.4 24 44 0.5
2001 1,232 15.4 938 454 5.7 25 33 0.4
2002 1,071 13.4 819 436 5.4 29 30 0.4
2003 1,132 14.1 865 428 5.3 22 34 0.4
2004 1,036 12.9 793 395 4.9 19 31 0.4
2005 983 12.3 745 378 4.7 24 21 0.3
2006 947 11.8 705 354 4.4 24 18 0.2
2007 909 11.4 707 379 4.7 9 16 0.2
2008 886 11.1 685 339 4.2 11 18 0.2
2009 757 9.5 539 281 3.5 9 25 0.3
2010 705 8.6 511 265 3.2 11 26 0.3
2011 684 8.4 501 264 3.2 16 32 0.4
2012 652 8.0 495 271 3.3 18 15 0.2
2013 650 8.0 473 258 3.2 7 17 0.2
2014 582 7.1 454 243 3.0 10 31 0.4
2015 575 7.0 444 240 2.9 5 20 0.2
2016 556 6.8 448 225 2.8 11 21 0.2
2017 608 7. 4 504 261 3.2 15 15 0.2
2018 553 6.8 421 230 2.8 13 Not available Not available
2019 566 6.9 463 253 3.1 11 Not available Not available
36
>> HISTORICAL TUBERCULOSIS DATA

NYC HEALTH DEPARTMENT EPI DATA BRIEFS
Epi Data Briefs are short
publications that highlight
data from Health Department
programs and projects. To
access recently published
reports, visit nyc.gov/health
and search for epi data.
"GET TESTED"/"GET TREATED" POSTERS
These 11x17 posters
highlight the benets of
TB testing and encourage
evaluation and treatment
for symptoms of TB
disease. Available
in Chinese.
RESOURCES FOR PROVIDERS AND THE PUBLIC
The Health Department offers a selection of culturally, technically and linguistically tailored TB education
materials and other resources for patients, the general public and health care providers. To access these
materials, policy updates and other information about TB and TB services in NYC, visit nyc.gov/health and
search for tuberculosis or call 311. For updates on TB related research, guidelines and events, please sign
TUBERCULOSIS
(TB)
TAKING CONTROL OF YOUR
What to Expect and How to Stay Healthy
"YOU CAN STOP TB" EDUCATIONAL POSTERS
This 11x17 poster provides
basic TB information
and includes illustrations
with captions. Available in
English, Spanish, French,
Haitian Creole, Hindu, Urdu,
Bengali, Tibetan, Tagalog and
Chinese; available in hard
copy or digital formats.
CLINICAL POLICIES AND PROTOCOLS
4th Edition.
Describes policies, protocols
and recommendations for the
prevention, treatment and
management of TB. The 5th
Edition will be available in 2020.
PATIENT BROCHURE
Taking Control of Your
Tuberculosis (TB): What to
Expect and How to Stay Healthy
General information for patients
starting treatment for latent TB
infection or active TB disease.
Available in 18 languages.
NYC NEIGHBORHOOD HEALTH ATLAS
This online resource provides
data on measures related to
social factors and health for
188 neighborhoods. To
access, visit nyc.gov/health
and search for health atlas.
>> NYC DATA RESOURCES ARE AVAILABLE ONLINE
37 New York City Health Department Annual Tuberculosis Summary, 2019
>> ADDITIONAL RESOURCES
TECHNICAL NOTES
• Data for 2019 are preliminary and reect the most complete information available as of Jan. 15, 2020.
• Data prior to 2019 have been updated since the release of the 2018 report. Data for these years reect the nal
numbers and may differ from ofcial estimates presented in previous reports.
• TB became a reportable disease on Jan. 19, 1897. From 1920-1940, only cases of pulmonary TB were reportable.
Beginning in 1978 the TB case denition was amended to consider people who had veried TB disease 12 or more
months before their current diagnosis as incident cases of TB disease.
• Data on patient sex are currently collected and categorized as male, female and transgender. In future reports,
more expansive categories of gender identity will be presented to reect changes in data collection.
• Age groupings have been changed from previous reports; as a result, count data for earlier years may differ from
previous reports.
• In all tables presenting data by birth in the U.S, column sums may not equal applicable totals due to missing or
unknown data.
• In all tables where data are presented by geography, column sums may not equal applicable totals due to missing
or unknown data.
• The sum of proportions do not always equal 100% due to rounding.
• All rates presented in this report are calculated per 100,000 population. Reported rates for earlier years may differ
from previous reports due to corrected data and changes in the denominators used to calculate rates. The sources
of denominator data are indicated throughout the report.
• The Health Department calculates population estimates based on modied U.S. Census Bureau interpolated
intercensal estimates. Data are modied to account for population undercounts in northwest Queens and southern
Brooklyn because of erroneously deleted housing units and housing units mislabeled as vacant. Population
estimates are updated as new data become available. Therefore, rates may differ from previously reported rates.
• U.S.-born refers to patients born in the 50 states, District of Columbia or other U.S. territories and outlying areas,
including American Samoa, Baker Island, Guam, Howland Island, Jarvis Island, Johnston Atoll, Kingman Reef,
Midway Island, Navassa Island, Northern Mariana Islands, Palmyra Atoll, Puerto Rico, U.S. Minor Outlying Islands,
U.S. Pacic Islands, Virgin Islands and Wake Island. All others with a known country of birth are considered
non-U.S.-born.
• Area-based poverty is dened using patients’ ZIP code of residence at the time of TB diagnosis. Poverty level by
ZIP code is based on the most recent American Community Survey ve-year sample data from the American
Community Survey that measures the proportion of residents living below the federal poverty level in that census
tract. The federal poverty level is a measure of income used by the U.S. government to determine eligibility for
subsidies, programs and benets. The Department of Health and Human Services updates the poverty guidelines
each January. Patients with addresses outside of NYC, addresses unable to be geocoded to a ZIP code or located
in ZIP codes where poverty level could not be determined were not assigned to a poverty level.
• The denition of excessive alcohol use has been changed from previous reports. The current denition of excessive
alcohol use is based on national denitions of binge drinking and heavy alcohol use from the Substance Abuse and
Mental Health Services Administration.
• The geographic distribution of cases is presented by the 42 United Hospital Fund neighborhoods. These
neighborhoods consist of adjoining ZIP codes that approximate NYC Community Planning Districts and contain
an average of 200,000 individuals.
• Data presented on HIV status reect information as collected by the Health Department. Misclassication of HIV
status may occur if a patient refused to disclose known status or refused to be tested for HIV while under care for
TB disease.
• Data on TB deaths are obtained from the NYC Ofce of Vital Statistics. Deaths recorded in a given year may include
cases diagnosed in a previous year.
• Product names are provided for identication purposes only; their use does not imply endorsement by the Health
Department.
38 New York City Health Department Annual Tuberculosis Summary, 2019
>> TECHNICAL NOTES

13
THE HEALTH DEPARTMENT PROVIDES A VARIETY OF
TB DIAGNOSTIC SERVICES, INCLUDING:
• Testing for TB infection using the latest
generation blood-based QuantiFERON®-TB
Gold test and TST
• Sputum induction
• Chest radiographs
• Medical evaluation
• Treatment for TB disease and LTBI
• DOT services, including vDOT
ADDITIONAL CLINICAL SERVICES PROVIDED AT EACH
CHEST CLINIC INCLUDE:
• Outpatient medical and nursing care
• Phlebotomy services
• Social services referrals
• HIV education and testing regardless of person’s
need for TB care
• TB evaluation for newly arrived immigrants and
refugees referred by the CDC
To make an appointment or to refer a patient, call the individual chest clinic or 311.
Eligible patients can be referred to one of four Health Department chest clinics located throughout NYC
for TB testing, radiography, sputum induction and treatment as needed. All chest clinic services, including
medication, are provided at no cost to the patient and regardless of immigration or insurance status.
295 Flatbush Ave. Ext.,
Fourth Floor
Brooklyn, NY 11201
718-249-1468
FORT GREENE
600 W. 168
th
St.,
Third Floor
New York, NY 10032
212-368-4500
WASHINGTON HEIGHTS
34-33 Junction Blvd.,
Second Floor
Queens, NY 11372
718-396-5134
CORONA
NEW YORK CITY HEALTH DEPARTMENT CHEST CLINICS
1309 Fulton Ave.,
First Floor
The Bronx, NY 10456
718-838-6876
MORRISANIA

